In the pharmaceutical industry, one term is increasingly taking centre stage these days: containment. It describes the process of enclosing a substance within a defined space, a method of product handling that is suitable for protecting operators and the environment when products are highly toxic and reactive.
At the same time, containment also prevents dusts, gases or vapours from being released into the environment and contaminating the product.
More than 60 per cent of pharmaceutical products are produced as tablets, capsules, coated tablets or similar. Active Pharmaceutical Ingredients(APIs) are becoming increasingly decisive in the effect of newer products in particular. In some markets, the market segment for High Potency Active Pharmaceutical Ingredients ( HPAPIs) is growing in the double-digit percentage range, mainly driven by oncological drugs.
The advantages of our containment applications
Best possible protection for employees
Protection against product contamination
Exclusion of environmental pollution
Use of proven components guarantees high Process Safety and stability - optimal basis for building containment components
Fast, convenient cleaning
Less cleaning of the premises enables quick shift changes and production restarts
Significantly reduced risk of cross-contamination
Significant savings through reduced or no use of air filters, ventilation or contaminated cleaning fluids

In addition to flexibility, our high containment applications focus on ease of use and safety for users and the environment.
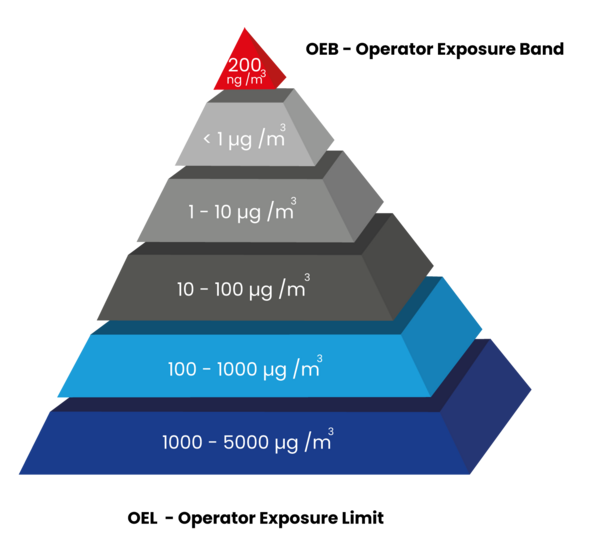
The globally standardised OEB classes (Operator Exposure Band) indicate the toxicity level of the substance on a containment scale.
OEL (Operator Exposure Limit), on the other hand, describes the average concentration of the active substance in the air at the workplace measured over a defined full stop of time at which no acute or chronic damage to the health of the operator is to be expected.
L.B. Bohle offers you a comprehensive range of state-of-the-art machines and systems for your tablet production. Individually tailored to your requirements, our machines optimise your production processes and ensure optimum quality. Our machine finder will guide you quickly and easily to the optimum solution for your processes.
With L.B. Bohle, you can achieve the highest production efficiency and maximum product quality.