Blogtext
Continuous and Digital – Contribution to the Subject of Continuous Manufacture and Process Monitoring
Nothing is more important in the production of pharmaceuticals than a precise workflow. Continuous procedures and digital systems secure constant quality of high-quality products and high patient safety. They ensure low costs at the same time.
No subject is discussed more intense in the pharmaceutical industry than introduction of continuous manufacture. As a pioneer and innovator in continuous manufacture, L.B. Bohle has set benchmarks in wet and dry granulation and coating since the 2000s. The Technology Center, inaugurated in 2015, is a unique testing and development center for continuous processes in pharmaceutical production.
Until now, however, a solution for continuous drying is still missing on the market. We at L.B. Bohle intended to fill this gap. In 2018, we presented the QbCon® 1, the first an R&D system that truly meets all requirements. Short residence times and narrow residence time distributions ensure reproducible quality. The drying process is so efficient that the residual moisture can remain below the initial moisture of the raw material.
Start of continuous manufacturing
QbCon® 1 is the ideal introduction to continuous manufacturing and has already proven its worth in production. Our development team has developed and optimized the solution in only six months. QbCon®1 is a system fully integrated into the software and mechanical system. It has various components such as powder dispensing, liquid dispensing, wet granulation and drying.
By using PAT, the production process can be tracked inline. This results in consistent and efficient production of high-quality products, high patient safety, lower costs, better process understanding and optimized process control.
The entire process is controlled and monitored via a touch panel. QbCon® 1 requires little space, is fast to install and achieves throughputs from 0.5 to 2.5 kg/h (nominal throughput).
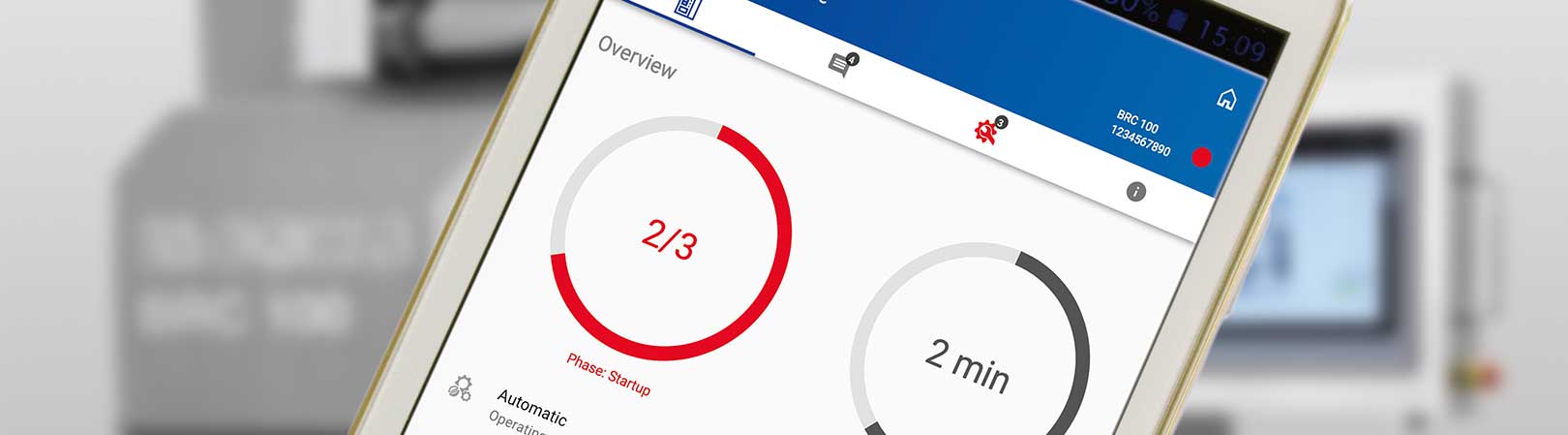
Monitoring app for pharmaceutical environment
As Managing Director Technology, I see a major challenge in avoiding machine downtimes and faulty production. Machine operators often have to keep a permanent eye on all process and operating details for entire production lines today. This is not always feasible with the large number of systems.
Therefore, we have developed a solution with which systems with programmable logic controllers (PLC) can send the recorded data to a communication server for processing. With our b.sure® app, the customer can keep an eye on his processes on authorized end devices or on his desktop PC at all times, in order to recognize and react to deviations from process parameters or errors on time.
In contrast to other monitoring solutions on the market that are based on an external cloud solution, b.sure® takes into account the special features of the highly sensitive pharmaceutical environment and processes the data in the customer’s network. Internal 5G networks will open up entirely new applications in automated manufacture of pharmaceutical products in future.
Ansprechpartner
Your contact for further information
Burkhard Schmidt
Sales Director
inquiry@lbbohle.de
+49 2524 – 93 23 0



